What Patients Should Know About the Stem Cell Harvesting and Banking Clinical Trial for RUNX1-FPD
Key Points
The Stem Cell Harvesting and Banking clinical trial is designed to explore the safety and feasibility of collecting and storing blood stem cells for potential future use.
Cells collected in this study are stored for that patient’s use only and are not part of a shared donor bank.
If technologies like gene editing advance, these stored stem cells may help enable new treatment approaches in the future for those who participate.
Individuals with RUNX1-FPD often ask whether there is anything they can do right now, given the risk of developing blood cancer in the future.
One area of research exploring this question is the Stem Cell Harvesting and Banking Clinical Trial, led at MD Anderson Cancer Center by Dr. Chitra Hosing and at the Children’s Hospital of Philadelphia by Dr. Tim Olson.
This multi-site effort focuses on determining whether stem cells can be safely collected from a patient today and stored for their own use in potential future therapies, including approaches such as gene editing that are still in development.
Why This Study Matters
Currently, the only established treatment for blood cancers associated with RUNX1-FPD is a stem cell transplant from a donor. While this approach can be effective, it carries risks and depends on finding a suitable donor.
This clinical trial is designed to answer two key questions:
Can stem cells be safely collected from individuals with RUNX1-FPD?
Can enough cells be collected for potential future treatment use?
The broader goal of this study is to prepare for a future in which new therapies, such as gene editing, may enable a RUNX1-FPD patient’s own stem cells to be corrected and used for treatment, thereby avoiding some of the risks of donor stem cell transplantation.
RUNX1 plays an essential role in how blood stem cells grow and develop, and fixing changes in this gene is more complex than it may seem. Unlike some blood disorders caused by a single common mutation, RUNX1-FPD can result from many different mutations, so there is no one-size-fits-all gene-editing solution.
In addition, people with RUNX1-FPD still have one working copy of the gene, so any therapy must be precise enough to avoid damaging that healthy copy. Current approaches also rely on treatments that can stress or damage blood stem cells, which may be especially sensitive in this condition.
Finally, to be effective, a large proportion of stem cells may need to be successfully corrected, and today’s technologies are still working toward that level of efficiency. Because of this, gene-editing approaches for RUNX1-FPD are still in early stages of research and are not yet available to patients.
Why Collect Stem Cells Now?
One of the key concepts behind this study is timing. Research suggests that stem cells collected earlier in life may be better suited for future use. This is because younger stem cells are more robust than older stem cells.
Furthermore, over time, cells can acquire additional genetic changes, which may affect their quality and function. This study is especially relevant for younger individuals, where earlier collection may help preserve healthier stem cells for potential future therapies.
What Participation Involves
Participation in the study involves several phases:
Screening: Before enrolling, participants complete a screening process to confirm eligibility and ensure their medical history suggests it is safe to proceed.
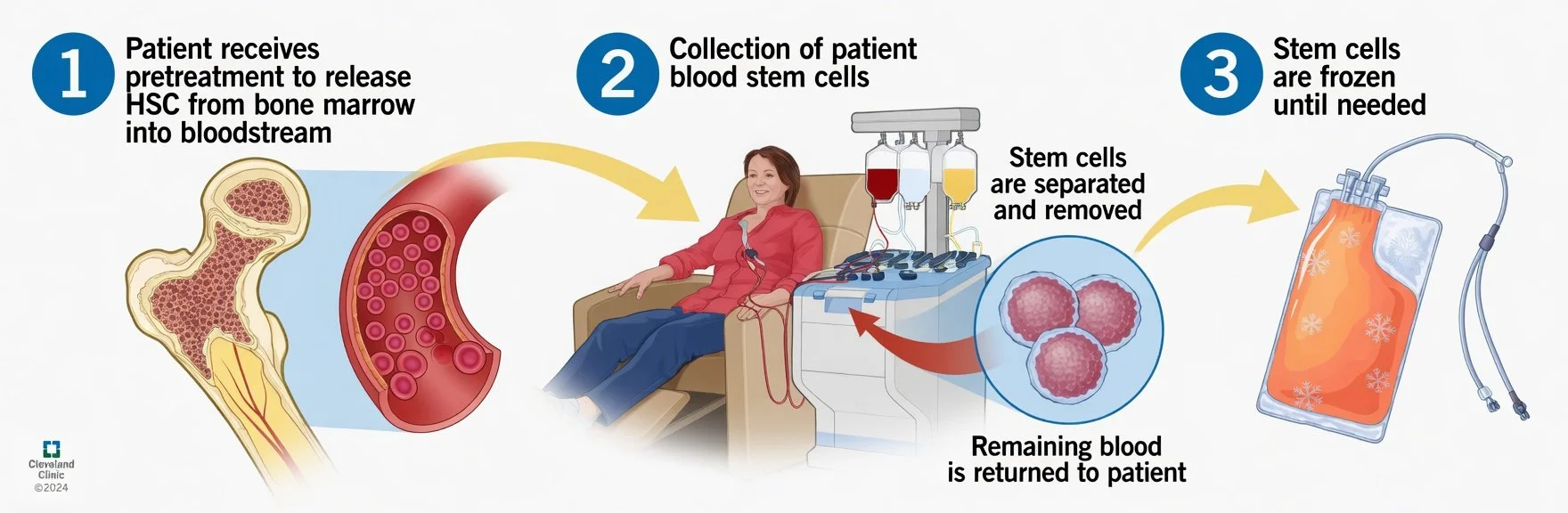
Mobilization: Participants receive daily single injections of a medication (G-CSF) over several days (typically 4–5 days). During this phase, patients may not come into the clinic at all and just self-inject. This medication helps move stem cells from the bone marrow into the bloodstream.
Stem Cell Collection: On day 5 or 6, depending on the number of stem cells doctors measure in the blood, stem cells are collected through a process called apheresis. During this procedure:
Blood is drawn from the body
A machine separates and collects stem cells from the blood
The remaining blood is returned to the body
The process typically takes a few hours and may be repeated on a second day if needed.
Most participants tolerate this process well, though some may experience temporary fatigue, lightheadedness, or tingling sensations. These side effects are typically temporary and manageable.
Follow-Up: Participants are monitored after collection to ensure safety, with a few follow-ups extending up to two years.
What Happens to the Stem Cells?
Once collected, the stem cells are processed and stored (or “banked”). They are divided into portions:
One portion may be reserved for a potential future auto-transplant
Another portion may support future gene therapy approaches
Any additional cells may be used to support ongoing research if the patient consents
These cells can be stored long-term, for decades. Whether or how these cells may be used in the future will depend on how new therapies develop over time.
Who Can Participate?
The study is currently enrolling adults first, with plans to expand to younger participants (ages 12–17) after initial safety is confirmed.
Some general considerations include:
Individuals who have already received a bone marrow transplant are not eligible
Patients with an existing blood cancer are excluded
Some individuals with certain acquired (non-inherited) somatic mutations in their bone marrow may still be eligible, depending on the type and extent
Participants may come from outside the United States, though insurance and logistical factors would need to be addressed
Additional eligibility criteria may apply, and the study team can help determine whether participation is appropriate for each individual.
Those interested in learning more can reach out to the study teams at MD Anderson or the Children’s Hospital of Philadelphia through the contacts listed at the bottom of this one-page study summary to:
Discuss eligibility
Review logistics, including travel and insurance
Ask questions about what to expect
Financial support from RRP may also be available for U.S.-based patients to help navigate participation. For more information, you can contact Dr. Amanda Eggen via email.
Looking Ahead
This clinical trial represents an important step toward expanding treatment possibilities for RUNX1-FPD. While there are still many unknowns, the study is focused on building a foundation that could help enable new approaches to care in the years ahead.
Common Questions
1. “I have a RUNX1 deletion, not a mutation. Does stem cell harvesting and banking still make sense for me?”
It’s true that today we do not yet have reliable gene-editing tools to correct large deletions such as those affecting some individuals in the RUNX1-FPD community. However, this is an area of very active research. Scientists around the world are working on new approaches, including advanced gene editing, gene replacement, and other cellular therapies. These new approaches may one day make it possible to address deletion-type variants.
Because of that, the inability to correct a deletion today should not be a reason to rule out stem cell collection and banking. Preserving your own healthy stem cells now may provide valuable options in the future as new therapies emerge. In other words, banking is a way of keeping doors open as science continues to advance.
2. “Could G-CSF (the medication) used for stem cell collection increase my risk of blood cancer?”
This is a common and important question. Based on decades of research, the answer is reassuring.
G-CSF has been widely used to help in the collection of stem cells from healthy donors for many years. Long-term follow-up studies, including large national donor registries, have not found an increased risk of blood cancers linked to short-term G-CSF use.
A major review of donor outcomes also found no convincing evidence that a short course of G-CSF raises the risk of developing a blood cancer.
Researchers have also studied whether G-CSF might affect clonal hematopoiesis, a process in which some blood cells develop genetic changes and begin to grow more than others, increasing blood cancer risk. So far, there is no clear evidence that short-term use of G-CSF causes these kinds of changes to expand.
Overall, current data show that G-CSF, when used briefly for stem cell collection, has a strong safety record and is not associated with an increased risk of blood cancer.
This blog is based on RRP-hosted webinar presentations by Dr. Chitra Hosing (MD Anderson Cancer Center) and Dr. Tim Olson (Children’s Hospital of Philadelphia).
References
de la Rubia J, Arbona C, del Cañizo MC, et al. Follow-up of healthy donors receiving granulocyte colony-stimulating factor for peripheral blood stem cell mobilization: a Spanish registry study. Haematologica. 2008. DOI: 10.3324/haematol.12285
Martino M, Console G, Dattola A, Callea I, Messina G, Moscato T, Massara E, Irrera G, Fedele R, Gervasi A, Bresolin G, Iacopino P. Short and long-term safety of lenograstim administration in healthy peripheral haematopoietic progenitor cell donors: a single centre experience. Bone Marrow Transplant. 2009 Aug;44(3):163-8. doi: 10.1038/bmt.2008.440. Epub 2009 Feb 2. PMID: 19182833.
Shaw, B., Confer, D., Hwang, W. et al. A review of the genetic and long-term effects of G-CSF injections in healthy donors: a reassuring lack of evidence for the development of haematological malignancies. Bone Marrow Transplant 50, 334–340 (2015). https://doi.org/10.1038/bmt.2014.278